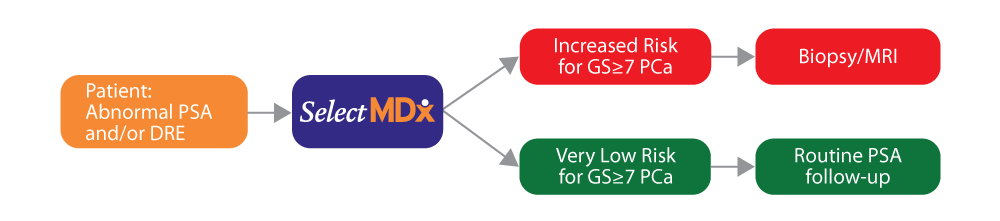
University Urology Associates Designated as a Center of Excellence for Rezūm
TM Water Vapor Therapy, a Treatment for Benign Prostatic Hyperplasia (BPH), or Enlarged Prostate.
Center of Excellence Recognizes High Degree of Experience with New Technology

University Urology Associates (UUA) has been designated as a Center of Excellence for RezūmTM Water Vapor Therapy treatment – a minimally invasive in-office procedure for men looking to treat their benign prostatic hyperplasia (BPH), not just the symptoms. Rezūm Therapy uses natural water vapor to reduce the size of the prostate and provide lasting symptom relief from BPH without invasive surgery or some of the potential side effects of prescription drugs.1 The Center of Excellence designation recognizes UUA’s high degree of experience with Rezūm Water Vapor Therapy and commitment to delivering significant, lasting symptom relief to men suffering from benign prostatic hyperplasia (BPH).1
BPH is a condition in which a man’s prostate enlarges and squeezes the urethra, causing frustrating symptoms such as frequent or urgent urination, a weak or interrupted urine stream, and the need to get up several times a night to urinate. It affects about 50% of men by age 60, and up to 90% of men by age 85.2
“Being designated as a Rezūm Center of Excellence is a meaningful recognition for us, as we strive to provide the best possible treatment of urological conditions for patients in the Greater Toronto Area and Ontario. For years, the primary options for treating BPH were through behavior modifications, medications, or invasive surgery. However, medications may not work for everyone and may have undesirable side effects such as dizziness, sexual dysfunction and lower sex drive.3 With Rezūm Therapy, we’re able to provide relief of BPH for men who do not want to take medication or want to avoid invasive surgery and implants.”
– Dr. Dean Elterman, University Urology Associates
During Rezūm Therapy, which takes place during one short office visit, natural water vapor is released throughout the targeted prostate tissue. When the steam contacts the tissue and turns back to water, it releases energy, killing the excess prostate cells that squeeze the urethra. Over time, the body’s natural healing response removes the dead cells and shrinks the prostate. With the extra tissue removed, the urethra opens, relieving BPH symptoms while preserving erectile and urinary function.1 The procedure does not require general anesthesia and is performed under oral sedation or local anesthesia.1
Rezūm Therapy has undergone extensive clinical trials to evaluate the safety and efficacy of the treatment.1 It is now included in the American Urological Association’s BPH treatment guidelines and has four-year follow-up data showing its ability to treat prostates with hyperplasia of the central zone, lateral lobe and/or median lobe.4
Rezūm Therapy is currently being used in many leading urology practices throughout the United States. To date, more than 55,000 patients around the world have been successfully treated with Rezūm Therapy. 5

1 McVary KT, Rogers T, Roehrborn CG. Rezūm water vapor thermal therapy for lower urinary tract symptoms associated with benign prostatic hyperplasia: 4-year results from randomized controlled study. Urology. 2019 Apr;126:171-9.
2 What is BPH? Urology Care Foundation website. https://www.urologyhealth.org/urologic-conditions/benign-prostatic-hyperplasia-(bph). Accessed August 5, 2019.
3 Roger K and Gilling P. Fast Facts: Benign Prostatic Hyperplasia, 7th edition. Health Press. 2011.
4 McVary KT, Gange SN, Gittleman MC et al. Minimally invasive prostate convective water vapor energy (WAVE) ablation: A multicenter, randomized, controlled study for treatment of lower urinary tract symptoms secondary to benign prostatic hyperplasia. J Urol. 2016 May;195(5):1529-38.
5 Date on file with Boston Scientific.
* Results from case studies are not necessarily predictive of results in other cases. Results in other cases may vary.
IMPORTANT INFORMATION: These materials are intended to describe common clinical considerations and procedural steps for the use of referenced technologies but may not be appropriate for every patient or case. Decisions surrounding patient care depend on the physician’s professional judgment in consideration of all available information for the individual case.
Boston Scientific (BSC) does not promote or encourage the use of its devices outside their approved labeling. Case studies are not necessarily representative of clinical outcomes in all cases as individual results may vary.
Caution: U.S. Federal law restricts this device to sale by or on the order of a physician.
All images are the property of Boston Scientific. All trademarks are the property of their respective owners.
© 2019 Boston Scientific Corporation or its affiliates. All rights reserved. URO-703802-AA NOV 2019